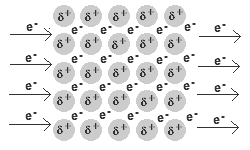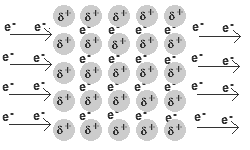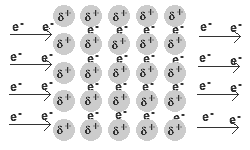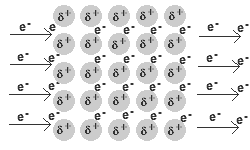
07.03 Metallic Bonds
Quiz by Kelly Gallagher
Feel free to use or edit a copy
includes Teacher and Student dashboards
Measure skillsfrom any curriculum
Measure skills
from any curriculum
Tag the questions with any skills you have. Your dashboard will track each student's mastery of each skill.
With a free account, teachers can
- edit the questions
- save a copy for later
- start a class game
- automatically assign follow-up activities based on students’ scores
- assign as homework
- share a link with colleagues
- print as a bubble sheet
8 questions
Show answers
- Q1What term means that metals can be pulled into thin strands or wires?ductilemalleableconductivityalloy30s
- Q2In metallic bonds, electrons are found in ___________.in the valence shellin the nucleus on the anionthe electron sea, surrounding positive cations30s
- Q3What term describes how metals can be rolled into sheets or beaten into complex shapes?alloymalleableconductiveductile30s
- Q4A(n) __________ is a material made of two or more elements that has the properties of metal.malleableconductivealloyduct30s
- Q5Metals ___________ electricity easily because electrons can move freely among the atoms.conductabsorbinsulateblock30s
- Q6Polished metals exhibit ____________ that is shiny. This is due to valence electrons.alloymalleabilityductilityluster30s
- Q7Melting and boiling points in metallic bonds are ___________extremely highextremelylow30s
- Q8Which of these metals is not an alloy?bronzesteelplatinumbrass30s