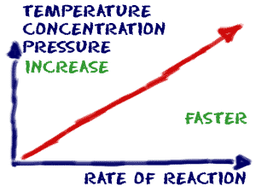
18.01 Factors Affecting Rate of Chemical Rxn
Quiz by Kelly Gallagher
Feel free to use or edit a copy
includes Teacher and Student dashboards
Measure skillsfrom any curriculum
Measure skills
from any curriculum
Tag the questions with any skills you have. Your dashboard will track each student's mastery of each skill.
With a free account, teachers can
- edit the questions
- save a copy for later
- start a class game
- automatically assign follow-up activities based on students’ scores
- assign as homework
- share a link with colleagues
- print as a bubble sheet
17 questions
Show answers
- Q1How quickly or slowly reactants turn into products is called the _________________.densitymetabolismAvogadro's raterate of reaction30s
- Q2Increasing the temperature causes atoms of reactants to move ___________.no different than at room temperaturemore quicklyslower30s
- Q3Decreasing the temperature of a reaction, causes atoms of reactants to move ______________.more qiucklymore slowlythe same as at room temperature30s
- Q4_________________ refers to how much solute is dissolved in a solution.molar massdensitymassconcentration30s
- Q5Increasing the concentration of a reactant ___________ your chances that a reaction will happen.does not affectincreasesdecreases30s
- Q6Decreasing the concentration of a reactant ___________ your chances that a reaction will happen.decreasesincreasesdoes not affect30s
- Q7___________ is the measure of how much area of an object is exposed.surface areadensitymassvolume30s
- Q8Increasing the exposed surface area ___________ your chances that a reaction will happen.increasesdecreasesdoes not affect30s
- Q9Decreasing the exposed surface area ___________ your chances that a reaction will happen.does not affectdecreasesincreases30s
- Q10A(n) __________ is a substance that speeds up the rate of a chemical reaction.enzymeprecipitatereactantcatalyst30s
- Q11The presence of a catalyst will __________ the rate of a chemical reaction.have no effect ondecreaseincrease30s
- Q12A catalyst speeds up the rate of a chemical reaction but _____________.evaporatescan also slow it downdoes not get used uprequires more energy30s
- Q13Decreasing the ________ will increase the rate of the reaction.size of the containerconcentrationtemperaturesurface area30s
- Q14__________ is a measure of the average kinetic energy of all of the particles in a sample of matter.concentrationtemperaturerate of reactionsurface area30s
- Q15______________ is the minimum energy colliding particles must have in order to react.activation energyreaction ratecollision theorykinetic momentum30s