CLOSER 3/20 Acid Base Chemistry 5h 5i
Quiz by Darrien James
Feel free to use or edit a copy
includes Teacher and Student dashboards
Measure skillsfrom any curriculum
Tag the questions with any skills you have. Your dashboard will track each student's mastery of each skill.
- edit the questions
- save a copy for later
- start a class game
- automatically assign follow-up activities based on students’ scores
- assign as homework
- share a link with colleagues
- print as a bubble sheet
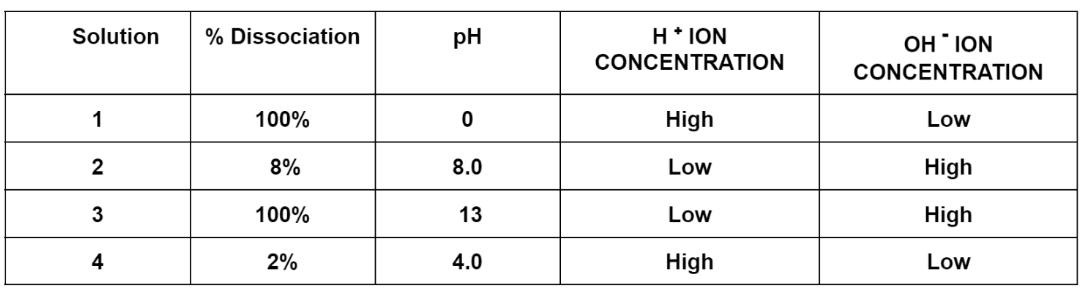
- Q1
A student is performing a neutralization reaction with 2 of the solutions shown in the table below. Which two solutions would be the BEST choice for their neutralization experiment?
Solutions 1 and 3 because they are a strong acid and a strong base.
Solutions 4 and 3 because they are a weak acid and a strong base.
Solutions 1 and 2 because they are a strong acid and a weak base.
Solutions 2 and 4 because they are a weak base and a weak acid.
30s - Q2
The properties below are listed for 4 substances:
Which of the following is the most acidic ?
B, because it has the highest H+ concentration
A, because it has the highest H+ concentration
C , because it has the highest H+ concentration
C , because it has the lowest H+ concentration
30s - Q3
Which substance has the lowest pH?
Substance B, because it has the highest concentration of hydronium, H+
Substance A, because it has the lowest concentration of hydronium, H+
Substance C, because it has the highest concentration of hydronium, H+
Substance A, because it has the highest concentration of hydronium, H+
30s - Q4
Which substance has the highest pH?
Substance A, because it has the highest concentration of hydronium, H+
Substance A, because it has the lowest concentration of hydronium, H+
Substance B, because it has the lowest concentration of hydronium, H+
Substance C, because it has the highest concentration of hydronium, H+
30s - Q5
The properties below are listed for 4 substances:
Which of the following is the most basic ?
B, because it has the highest H+ concentration
C , because it has the lowest H+ concentration
A, because it has the lowest H+ concentration
C , because it has the highest H+ concentration
30s