Solutions Exam Review AKS 5
Quiz by Darrien James
Feel free to use or edit a copy
includes Teacher and Student dashboards
Measure skillsfrom any curriculum
Tag the questions with any skills you have. Your dashboard will track each student's mastery of each skill.
- edit the questions
- save a copy for later
- start a class game
- automatically assign follow-up activities based on students’ scores
- assign as homework
- share a link with colleagues
- print as a bubble sheet
- Q1
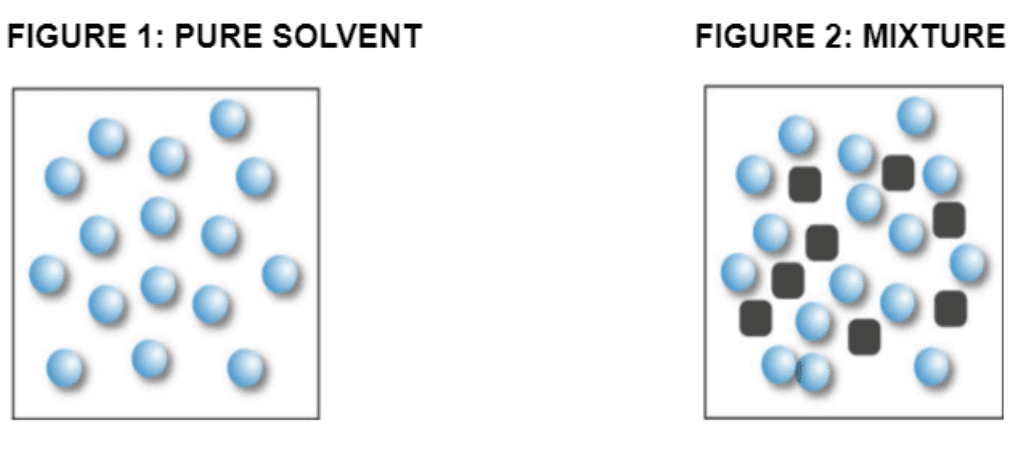
Examine the picture, which of the statements is TRUE regarding the solutions?
The pure solvent in figure 1 would freeze at a lower temperature than the mixture in figure 2.
The mixture in figure 2 would freeze at a lower temperature than the pure solvent in figure 1.
The pure solvent in figure 1 would boil at a higher temperature than the mixture in figure 2.
The mixture in figure 2 would boil at a lower temperature than the pure solvent in figure 1.
30s - Q2
What is the molarity of a solution of KCl if there are 74.55 grams of KCl dissolved in water to make 2.0 liters of solution?
150 M
0.5 M
0.0134 M
1.0 M
30s - Q3
What is the molarity of a solution of NaCl if there are 176 grams of NaCl dissolved in water to make 4.0 liters of solution?
704 M
3.0 M
0.023 M
0.75 M
30s - Q4
What is the molarity of a solution with 3.00 moles LiBr dissolved in enough water to make 500 mL of solution?
0.19 M
6.0 M
131 M
30 M
30s - Q5
What is the molarity of a solution with 4.00 moles CaSO4 dissolved in enough water to make 600 mL of solution?
6.67 M
2400 M
150 M
0.007 M
30s - Q6
What is the volume in liters of a solution that contains 10 moles MgCl2 in a 2 M MgCl2 solution?
20 L
5 L
0.2 L
0.25 L
30s - Q7
What is the volume in liters of a solution that contains 2.5 moles Al(OH)3 in a 3 M Al(OH)3 solution?
7.5 L
1.2 L
0.25 L
0.83 L
30s - Q8
What is the volume in liters of a solution that contains 3 moles Ca(NO3)2 in a 2 M Ca(NO3)2 solution?
15 L
0.67 L
1.5 L
6 L
30s - Q9
How many moles of NaOH are there in 2.0 liters of a 6 M NaOH solution?
16.4 mol
12 mol
1.73 mol
0.58 mol
30s - Q10
How many moles Li2S are there in 4.1 liters of a 7.1 M Li2S solution?
0.58 mol
16.4 mol
29.1 mol
1.73 mol
30s - Q11
How many moles AlBr3 are there in 9.0 liters of a 3 M AlBr3 solution?
6 mol
27 mol
0.33 mol
3 mol
30s - Q12
A 0.5 L solution of NaCl has a molarity 3 M. How many grams of NaCl were used to make this
aqueous solution?
58.44 g
1.5 g
87.7 g
6 g
30s - Q13
A 1.0 L solution of Co(NO3)2 has a molarity 2.0 M. How many grams of Co(NO3)2 were used to make this aqueous solution?
365.8 g
0.5 g
182.9 g
2.0 g
30s - Q14
Using the graph attached, at 30ºC, which of the following solutes is LEAST soluble in 100g H2O?
NaCl
KCl
KClO3
NaNO3
30s - Q15
Using the graph attached, at 20ºC, which of the following solutes is MOST soluble in 100g H2O?
KNO3
KCl
NaNO3
NaCl
30s